Here’s Beginning to See the Light by the Velvet Underground to almost the end of the semester and seeing hope!
What are HLA molecules?
Human Leukocyte Antigens (HLA) are proteins found on most cells in your body that help the immune system distinguish its allies from aliens. HLA class I molecules are found on almost all (except mature red blood cells) nucleated cells by forming a functional receptor, and HLA Class II molecules are primarily found on professional antigen presenting cells such as dendritic cells and B cells. These molecules display an array of peptides on their surface, which are small fragments of proteins processed within the cell. Class I molecules present endogenous antigens, which proteins derived from the cell itself, such as viral proteins are displayed. In contrast, Class II molecules present exogenous antigens that have been engulfed from outside the cell via endocytosis.
Both classes of cells serve to present antigens to T cells. Class I molecules consist of a heavy chain and a beta microglobulin, presenting antigens to CD8 and cytotoxic T cells to trigger the destruction of infected cells. Class II molecules are composed of two chains, alpha and beta, and they present antigens to CD4 and helper T cells to coordinate a broader immune response. Despite these differences, they are similar in that both are highly polymorphic, meaning there is extreme variation between individuals, making matching so difficult in the context of clinical transplantation.
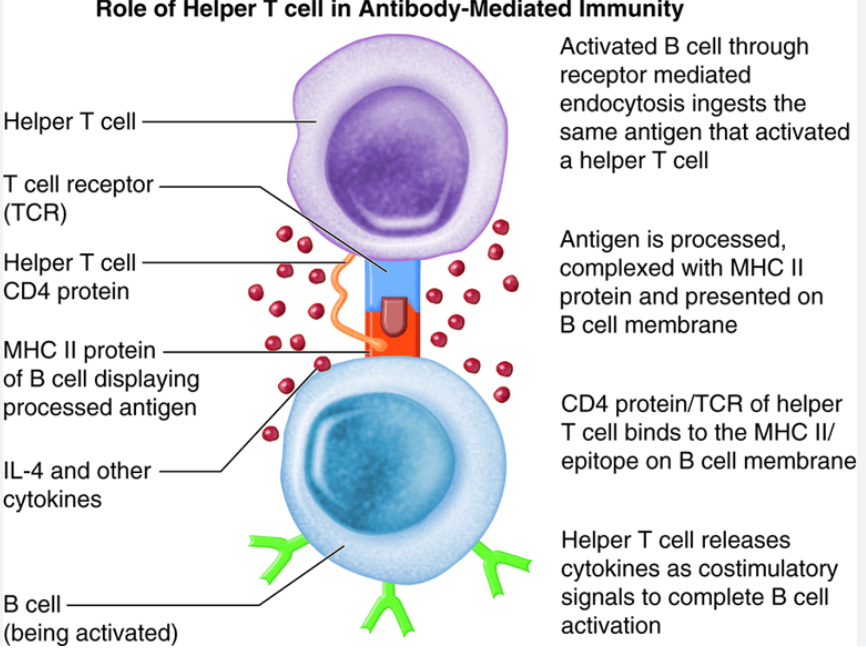
Photo credit Colorado Community College System
Role of HLA matching for solid organ transplants
Solid organ transplants are common for patients with end-stage organ failure, it is for multiple organs such as the kidneys, heart, liver, and lungs. HLA matching is a process used to determine how closely a donor’s tissue markers match a recipient’s. While we might usually think that this compatibility is important for bone marrow transplants, but in solid organ transplantation is has more various roles. The primary HLA markers targeted for matching are HLA-A, -B, and -DR, with HLA-DR often identified as the most critical locus for preventing early rejection. For kidney transplants, precise HLA matching is a priority because it significantly extends long-term survival and reduces the need for immunosuppression. In contrast, the liver is uniquely resilient and can often be successfully transplanted with a high degree of HLA mismatch compared to other organs.
Individuals who receive a solid organ transplant must remain on immunosuppressive (anti-rejection) drugs for the rest of their lives. These medications work by dampening the immune system’s natural urge to attack the foreign tissue. Without these drugs, the recipient’s T cells would eventually recognize the donor HLA molecules as foreign and initiate a rejection. Even years after a successful surgery when people might think the organ would fit in with the body more, one wrong dose of medication can trigger an immune response that damages the organ entirely. Therefore, lifelong adherence to a complex medication regimen is the standard of care for all solid organ recipients.
HLA compatibility in bone marrow transplants
In bone marrow or hematopoietic stem cell transplantation, the precision of HLA matching is much more rigorous than in solid organ surgery. A 10/10 match refers to perfect compatibility of 5 HLA marker locations (HLA-A, -B, -C, -DRB1, and -DQB1). A haploidentical match occurs when the donor matches exactly half of the recipient’s HLA markers, which is common between parents and children. 10/10 matches are generally considered better as it minimizes the risk of Graft-versus-Host Disease (GVHD), where the donor’s new immune cells attack the recipient’s healthy tissues. In GVHD, HLA mismatches makes the graft sees the host as foreign, possibly leading to severe inflammation.
Bone marrow patients who achieve successful engraftment and immune tolerance can often be weaned off anti-rejection drugs after six to twelve months. The risk of GVHD is the primary reason why HLA compatibility is more stringent in bone marrow transplants than in solid organ transplants, as in a solid organ transplant the recipient’s immune system attacks the organ, but in bone marrow transplants, the new immune system attacks the entire body of the recipient. Therefore, this inverted immune response means that even minor mismatches can trigger a massive systemic inflammatory response. However, if the transplant is successful, the patient would possess this new immune system that views the host body as self. This transition allows many patients to eventually stop taking immunosuppressants entirely.
