Hello to March! Here’s some great classic spring music, Here There and Everywhere by the Common People, a high school who did fun music band back then. The song was orginally by the Beatles.
NDM-1 plasmid and antibiotic resistance
NDM-1 refers to New Delhi metallo beta lactamase-1, a novel MBL that confers resistance to all β-lactam antibiotics. It was first discovered in Sweden in a K. pneumoniae strain isolated from a patient from New Delhi, India in 2008, and some retrospective studies identified NDM-1 from 2006. Since its discovery, it has become endemic to the Balkans and Middle East, spreading to the world as it has the ability to spread rapidly between different bacterial strains, such as in E. coli. The gene that encodes for NDM-1 is blaNDM-1, and it spreads to other bacteria by horizontal gene transfer, specifically conjugation, unidirectionally from a donor bacterium to a recipient bacterium through direct contact. Efficient transfer also envolves novel hybrid plasmids formed through recombination.
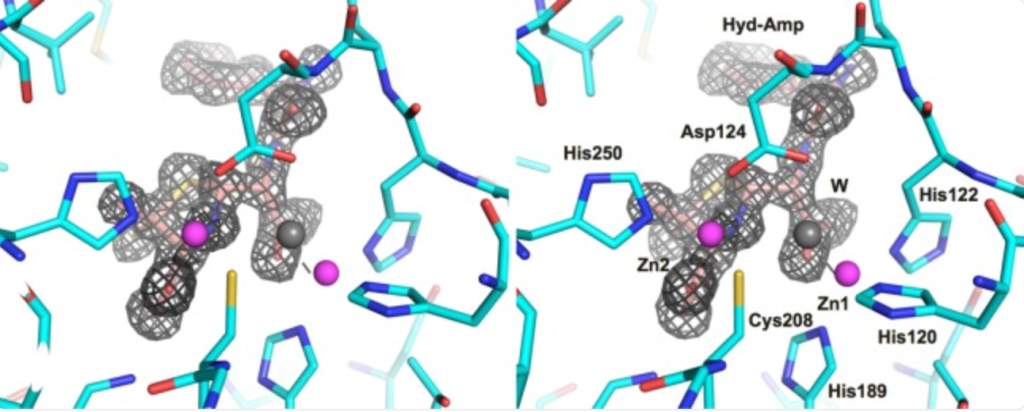
NDM-1 in complex with hydrolyzed ampicillin. Image credit Kim et al.
The plasmid is most commonly found in Gram-negative bacteria that naturally inhabit the human gut, making household and hospital settings very susceptible to transmission. Between 2019 and 2023, NDM-CRE infections surged by more than 460% in the United States (CRE meaning carbapenem-resistant Enterobacterales, resistant to some of the strongest antibiotics available). In a New York City report, cases increased from 58 in 2019 to 388 in 2024. This suggests a high possibility of community transmission beyond healthcare settings where CRE transmission has previously been identified. These results show the need for alternate solutions than antibiotics for better infection control.
Human behaviors that contribute to the rise of antibiotic resistance
Antibiotic resistance is a global public health problem, as it doesn’t just affect you but the whole population. Specific types of bacteria adapt after constant use of antibiotics to treat bacterial infections, and we commonly use new drugs to treat the same disease to prevent this. Overprescribing antibiotics is the primary driver of rising bacterial resistance nowadays, and is estimated to have caused more than a million deaths worldwide in 2019. As bacteria cells frequently divide and multiply, at each division, there is the possibility of mutation that would bring out resistance genetics, so each time antibiotics are used, bacteria have another chance to adapt, leading to antibiotic resistance.
It might be common sense that stopping antibiotic courses early would help combat antibiotic resistance as we’re giving less antibiotics in general, but it might cause incomplete clearance of bacteria and recurrence of the disease, needing more antibiotics in total. Agricultural use would also lead to antibiotic resistance, as veterinary antibiotics would pre-select the resistant bacteria. These bacteria could also spread to humans through food, water, and direct contact, making treatments less effective. Global travel acts as a vector for spreading antimicrobial-resistant bacteria, with roughly 30% of international travelers returning with antibiotic-resistant pathogens. Through very easy travel routes such as direct international flights, not only humans but also animals and goods could carry antibiotic-resistant pathogens.
Overprescribing antibiotics in early childhood
Overprescribing antibiotics in early childhood could cause life health issues with kids growing up. According to a study, children exposed to antibiotics before age 2 face a 24% higher risk of developing asthma. This risk appears to follow a “dose-response” pattern, meaning the probability of respiratory issues increases with every subsequent course of medication. In the gastrointestinal system, frequent antibiotic use is associated with a 42% increased risk of Celiac Disease Autoimmunity. These findings are related to the Hygiene Hypothesis, as it suggests that reduced early childhood exposure to microbes, bacteria, and parasites, prevents the immune system from developing proper tolerance, leading to increased allergies and autoimmune disease. Consequently, the medical community is increasingly viewing the first 1,000 days of life as a “critical window” where antibiotic stewardship is most vital.
A popular view is that antibiotics in early life would lead to spectrum disorders such as ADHD and autism as children exposed to antibiotics were 18% more likely to later be diagnosed with ADHD. There are many cases against this, such as research proof that antibiotics at 1 to < 3 years did not show an association with IBD or Crohn’s disease. There is also a sibling-matched analyses in 2024 that challenged this. When comparing siblings with different antibiotic histories, the correlation to autism largely disappeared, indicating that genetics or shared environments rather than drugs may be the true cause. This highlights the complexity of our gut brain axis and the need for more studies to separate correlation from causation in medical areas.
